
INTRODUCTION
Any medical student would agree that one of the most pressing issues that drives most of us to tears of desperation is “How on earth do we commit so much factual information into our memory?!” So after yet another unsuccessful attempt at rote learning the tongue twisting names of hundreds of drugs for an upcoming pharmacology exam I was left wishing for a technique to simply zap my brain with all this information and form memories at the speed of light so that I could give my driven to exhaustion brain, some much needed rest. So after the exam I did some research on memories and how they are formed and was simply thrilled at the immense advancements that neuroscientists have made in the field of memory, so much so that today it has become possible to artificially create memories in the brain not by some mere psychological techniques but by actually tampering with the brains physical molecular framework. In what may seem reminiscent of science fiction movies like Total Recall, Inception and The Eternal Sunshine of the Spotless Mind, scientists have now managed to alter existing memories to create false memories in mice and even have managed to create an entirely fictional memory in the brains of mice, but before I get to this amazing breakthrough I would like to start by giving you a brief insight into memory and how it is formed and stored in the brain and the techniques used to identify such memory traces in order to alter them.
HISTORICAL PERSPECTIVE
Richard Semon was the first person to identify almost a century back in 1921 that memories were formed due to a synchronous activation of groups of specific neuron in a pattern just akin to how a specific circuit encoded in a computer executes different functions. He coined the term ‘engram’ for these enduring yet primarily latent modifications in the irritable substance of the brain and also came up with yet another term ‘ecphory’ which was the supposed reactivation of a latent memory engram by an external cue or in other words the act of recalling or retrieving an already encoded memory in the brain. After this came the obvious and all important search for the circuit of neurons that form an engram in the brain. Karl Lashley pioneered a set of experiments where mice were tasked with a maze task and he attempted to see how lesions introduced at various parts of the brain impacted the performance of the mice in successfully completing the task. He was however unsuccessful in determining any one distinct portion of the brain as the source of memory engram cell and instead concluded that the memory engram cells were widely spread through the cerebral cortex and the extent of performance impairment depends directly on the size of the lesion introduced. His discovery was succeeded by that Penfield and Rasmussen who attributed a major chunk of episodic memory formation process to the Medial Temporal Lobe. This finding was supported by clinical evidence by Scoville and Milner who found that H.M. a patient who had his MTL surgically resected suffering from severe anterograde amnesia and graded retrograde amnesia. As far as the physical nature of engrams are concerned the guiding principle has been Donald Hebb who stated that ‘neurons that fire together, wire together’ which posits that the neurons that are simultaneously undergoing activation as part of an engram during the encoding of a memory undergo synaptic changes that strengthen the association between these neurons which was also supported by the Long Term Potentiation theory put forward by Bliss and Lomo. Previously studies have managed to identify neuronal sub populations that are part of a memory engram circuit by correlational evidence(Reijmer’s et al) and by loss of function evidence(Han et al) however the most substantial proof that a group of neurons belong to a memory engram must come from a gain of function study. This is if a selected group of neurons being artificially activated leads to the recall of that memory and produced a physical response as a result of the recall. However the major problem here arises from the technical challenges associated with identifying the selected neuronal members of an engram circuit and distinguishing them from their seemingly indistinguishable neighbours- a situation akin to finding a needle in a haystack.
OPTOGENETICS AND THE TETRACYCLINE METHOD OF REGULATED GENE EXPRESSION

After this brief insight onto the major milestones in the research of memory and engrams I shall now delve into details of a novel method that has taken the world of neuroscience by storm. Earlier the main techniques used to stimulate neurons were primarily of two types. The first method was chemical by employing pharmacological agents however this took too long and was not suitable for targeted delivery to a small subsection of neurons due to the considerable overlap between neuronal membrane receptors and synapses in the complicated architecture of the brain. Yet another approach was using electricity as a stimulus which was faster no doubt but still couldn’t achieve the targeted delivery that was desired to selectively activate the memory engram cells. This was when a novel technique came into the scene- optogenetics. The discovery of optogenetics was when it was observed that certain green algae were photosensitive and swam in a direction opposite to that of a light beam. From these algae were isolated photosensitive ion channels called channelrhodopsins and these form the basis of the technique of optogenetics.
So now the question arises that how do we identify the subsets of neurons that were active during the formation of a particular memory. In order to do so scientists make use of IEG’s or Immediately Early Genes that have a very short half life and are expressed in response to a high level of neuronal firing. (Sagar et al). The three most commonly used genes are arc, cfos and zif268 and they provide a record of neuronal activity rather a map of neurons that were activated a few hours prior.(Guzowski et al). The next step needed was to achieve an activity dependant marking of the genes that were active during the formation of a memory ( those that show a high expression of IEGs) by combining cfos promoter with the tetracycline system for regulation of gene expression. (Reijmers et al).
Tetracycline controlled gene expression was derived from the method used by gram negative bacteria to acquire tetracycline resistance. There are two systems in this technique know as Tet off and Tet on systems depending on how the cell responds to the use of a tetracycline like Doxycycline.
The Tet off system uses the Tta protein created by fusing TetR also known as the tetracycline repressor which is found in the bacterium Escherichia Coli with the activation domain of VP16 found in Herpes Simplex Virus. This Tta binds to the TetO promoter region. Several such TetO sequence repeats are seen just upstream of a minimal promoter eg CMV promoter. The entire set of TetO sequence repeats with the minimal promoter form the Tetracycline Response Element (TRE). This element responds to the binding of the Tta protein by causing an increase in the expression of genes downstream to the TRE. In case of a Tet off system the antibiotic tetracycline or doxycycline binds to the Tta and represses it thereby preventing its binding to the TRE causing decreased expression of the genes selected. In the case of a Tet On system the contrary is true as the rTta protein is only equipped to bind to the TRE if it is concurrently bound to the tetracycline molecule.
So with this basic general overview of the techniques used to label selective populations of neurons I shall now strive to explain in detail how these techniques were used for the first time in order to artificially create a fear memory in the brain of transgenic mice.
INCEPTION OF A FALSE MEMORY BY OPTOGENETIC MANIPULATION OF A HIPPOCAMPAL MEMORY ENGRAM.
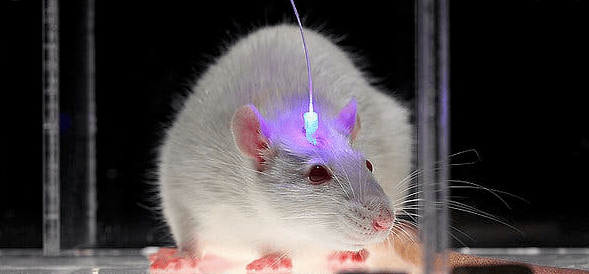
In this study the test subjects used were c-fos tTa transgenic mice, mice that were transgenically modified to express an allele that expresses the tTa under the control of the cfos promoter. c fos as mentioned earlier is one of the three most commonly used IEG’s and in neurons that are active the cfos promoter results in transcription of the doxycycline sensitive tTa protein which also shares the same promoter. Hence in the absence of doxycycline the tTa is permitted to bind to the TRE that results in expression of the downstream genes.
These mice were then injected with a virus called AAV9-TRE-ChR2-EYFP virus near their dentate gyrus. So when the neurons are active in the absence of doxycycline the tTa which is expressed due to the cfos promoter being active binds to the TRE causing increased expression of the ChR2( channel rhodopsin- a light sensitive channel) EYFP( enhanced yellow fluorescent protein) during the defined time period that the mice are kept off doxycycline. These labelled neurons can then be selectively reactivated by simply shining light through an optical fibre cable targeting the dorsal DG introduced into the cranium of the mouse.
So the first test done was to check whether activating the neurons that were active during a fear response by shining light was sufficient to reinstate that particular fear memory in the absence of any external fear stimulus.
So one group of mice were put on doxycycline and then underwent a habituation period in a context A where their basic levels of freezing (the classical response shown by mice to a fearful stimulus)were recorded during both light on and light off epochs. After this they were taken off doxycycline for three days and then underwent fear conditioning by delivering a foot shock in a different context B. After which they were once again started on doxycycline in order to prevent any labelling of any further neurons other than those that were active during the fear conditioning period in context B. Then the mice were once again taken to context A and the levels of freezing were noted during both light on and light off epochs and it was found that the levels were considerably higher during the light on epoch as compared to prior to the fear conditioning when there was not much significant difference between the two epochs. Also those mice (NS group) that underwent the very same protocol with the only difference being that they did not receive a foot shock also didn’t show any increased freezing during light on epochs.
Another group of mice (EYFP) that were injected with a virus (AAV9-TRE-EYFP) that didn’t have a gene for channel rhodopsin 2 also didn’t show any increase in freezing levels during light on epochs. This conclusively proved that the increased levels of freezing shown during light on epochs by the mice in the first group was due to the optogenetic reactivation of the memory engram neurons of the DG that were specific to that particular fear memory of receiving a foot shock. In addition it was also seen that the level of freezing during light on epochs were significantly higher when the duration that the mice were off doxycycline was reduced to just one day probably due to reduction in the labelling of unwanted competitive non contextual neurons that were active during the longer period of time that the mice were off doxycycline. In addition the levels of freezing were significantly higher in those who underwent optogenetic reactivation of neurons of bilateral hippocampus systems compared to unilateral reactivation. Figures 11-a
The overall results of this experiment was sufficient to conclude that the DG cells that were active during the memory encoding showed expression of cfos and hence were labelled with ChR2 and EYFP and defined a population of neurons whose activation by light was sufficient for the recall of that specific fear memory.
After this trial the researchers went on a step further to see if they could form an artificial Conditioned Stimulus(CS) and Unconditioned Stimulus(US) association what they referred to as a false memory using these same techniques.
In this test they took virus injected and fibre optic cable implanted mice off doxycycline and allowed them to explore a context A during which the active neurons that were responsible for forming a memory of context A were labelled with ChR2- mcherry. Then these mice underwent fear conditioning by means of a foot shock in a different context B during which the neurons that formed a part of the memory engram for context A were optically reactivated by switching on the light. If the light reactivated cells of context a were sufficient to serve as a functional CS for the US of a foot shock given in context B then the mice would show a false memory response by having significantly higher levels of freezing in context A as compared to a totally different context C and amazingly that’s exactly what was seen- in other words a false memory of a sort had been successfully created for the first time using techniques that directly manipulate the physical substance of the brain and not merely by psychological association tricks as had been used in the past to create false memories. This result wasn’t a mere generalisation as was confirmed by the fact that those cells injected with the virus AAV9-mcherry that did not express ChR2 didn’t show any significantly higher levels of freezing in context A as compared to C.
The researchers had done this experiment by labelling cells in the DG however the same results couldn’t be reproduced while labelling active cells of the CA1 region. This was hypothesised because of a greater degree of overlap seen between different contexts in the active neurons which was as high as 50% in the case of CA1 while only about 6% in the case of the DG. Hence the formation of a distinct memory that was easily distinguishable among different contexts was an issue for the case of the CA1.
Till now it had been shown that the mice froze more in context A when physically kept in that context. Now what was left to be tested was that did the mice freeze when the labelled neurons for the context A were simply activated by light or was an external exposure to A necessary to elicit the freezing response and much to the satisfaction of the scientists even a mere switching on of the light onto the labelled neurons when the mice were in a totally different context D was sufficient to cause increased levels of freezing(by 25%) as compared to the mice that were only labelled with mcherry or those that did not receive any light during the fear conditioning in context B or in those whose CA1 region was manipulated instead of the DG. This is sufficient evidence to prove that the memory was indeed a real one and could be reactivated by stimulation just as any other memory that had been formed physiologically.
Further solid proof that a lasting memory had been created was to histologically map the levels of cfos expression in downstream region of the fear memory recall circuit- namely the basolateral and central amygdala and lo and behold there was a significantly higher level of cfos expression in both the false memory context A as well as the natural fear conditioning context B as compared to a control group in a neutral context showing that undoubtedly the circuits involved in the recall of the falsely generated fear memory was extremely similar to the ones involved in the recall of a genuine naturally formed fear memory.
The awe inspiring results of this experiment opened up new avenues for various possibilities. The potential of this technology is immense and can be of great significance in the treatment of disorders as far ranging as depression and PTSD to neurodegenerative disorders like Alzheimer’s.
ACTIVATING POSITIVE MEMORY ENGRAMS SUPPRESSES DEPRESSION LIKE BEHAVIOUR
Hence as is the natural course of events the next set of experiments aimed to see if an already formed memory could be tampered with to change the experience to a positive one.
Depression a psychiatric disorder that affects a majority of today’s population has been traditionally treated with pharmacological agents which over long periods of time does produce significant side effects that markedly impair the quality of living of the patients. Hence in order to see whether this new technique of optogenetically manipulating neurons could pass as a possible treatment option for depressive disorders yet another set of experiments were conducted.
Well as far as emotions go it seems that mice aren’t all that different form human beings for funnily enough just like human beings even for mice exposure to mice of the opposite gender constituted an extremely positive memory experience. During the time that the male mice were exposed to female mice they were taken off doxycycline to allow labelling of neurons specific to the positive memory of the female mice by the channel rhodopsins. Then these mice were subjected to a Chronic Immobilisation Stress (CIS) of 10 days duration following which their performance on various tests such as the OFT( Open field test) and the EPMT(Elevated Maze Plus Test) for detecting anxiety like behaviours and the Tail Suspension Test(TST) as a measure of active or passive escape behaviour in response to a stressful situation and the SPT(Sucrose Preference Test) to check for annedonia(inability to experience pleasure). The response to all these tests were significantly decreased in those mice that were exposed to CIS as compared to the controls that were not. What was exciting was that the optogenetic reactivation of the positive memory caused an improved performance in these tests in the stressed animals while there seemed to be not much difference in the performance of controls who weren’t exposed to CIS or in those mice that were only labelled with mcherry. A subsequent measurement of cfos expression in various parts of the brain unveiled that the areas implicated in the rescue caused by the positive memory included mainly the amygdala and the nucleus accumbens both having significantly higher cfos activity.
However is this response short lived and dependant on the light being switched on or does the stimulation of these positive memories produce a lasting healing impact on the brain long after the light is switched off, without which there doesn’t seem to be much scope of this technique replacing the pharmacological therapeutic options was the next question to be answered. To investigate this the mice were kept with light on for varying periods of time and it was found that when the light was on for a period of no less that five days the response to the tests of this group was almost comparable to the completely unstressed mice group. What I felt was the most remarkable was that the negative effects of depression on neurogenesis in the hippocampus also seem to have been reversed by the chronic optogenetic activation of the positive memory trace as this group showed higher polysialated neuronal cell adhesion molecule(PSA-NCAM) and doublecortin(DCX) both of which are considered to be markers of increased neuronal biogenesis thus offering hope that this technology can even reverse the long lasting negative impacts of depression on cognition and memory. So the age old adage that love and positivity heals seem to be incredibly true in this context!
MEMORY FORMATION IN THE ABSENCE OF EXPERIENCE
Albeit all the above experiments seeming to be really thrilling and as close to sci fi as one can get it still doesn’t help solve my initial problem as a lazy medical student looking for an easy way out of hard work for I needed a technique to introduce a completely artificial memory of my pharmacology book into my brain without any external stimulus of any sort such as the books pages. Devious as this may seem I was simply overjoyed to see that a step indeed seems to have been taken in this direction.
Another group of researchers managed to create an entirely artificial memory without any external stimulus of any sort in the brain of mice.
I shall now describe in detail how this impressive feat was achieved.
To implant an artificial memory successfully two criteria had been proposed-
- the learning event should take place entirely intracranial by optogenetic methods(that is without me having to even catch a glimpse of my pharmacology text book)
- the presence of the implanted memory should be demonstrated via the presentation of a real external memory retrieval cue( the pharmacology question paper in my analogy)
However unfortunately since current research hasn’t advanced to levels that can correctly recognise such complex memories as is needed to remember pharmacological drugs for the context of this experiment the architecturally less complex olfactory glomerulus circuits were used as the CS while the US could be either having a positive valence(rewarding) or negative valence(aversive).
The odours chosen were acetophenone( a cherry blossom odour)or carvone (a spearmint odour)as these stimulated different OR (Odorant Receptor ) populations.
In this experiment the transgenic mice with OSN(Olfactory Sensory Neurons) expressing M72 OR (receptor that detects acetophenone but not carvone) along with ChR2 were used and the neurons whose receptors detected the cherry blossom odor of acetophenone were activated by light and spatially coupled with activation of the Lateral Habenula (LHb) inputs to the VTA(Ventral Tegmental Nucleus) which was shown by a previous study to mediate an aversive response. After this optogenetic activation when the mice were placed in a box with two ends – one emanating the cherry blossom odor and the other the spearmint odor the mouse scurried quickly towards the spearmint side avoiding the cherry blossom side due to it having formed a completely artificial aversive memory towards an odor it had never smelled before having associated it with a US of negative valence that it never experienced. In a parallel experiment stimulation of the LDT(Lateral Dorso tegmental nucleus) inputs to the VTA which led to a rewarding response being paired with optic stimulation of the M72 OR associated with cherry blossom acetophenone odour caused the mice to scurry towards the acetophenone side in favor of the spearmint carvone side even though it wasn’t as if a female mouse or a huge slice of cheese had awaited them in association with this cherry blossom odour that it had never even smelt prior to this. Amazing isn’t it?!!!
Also training naive mice didn’t show any natural preference or aversion to any of the odours, also mice that underwent the optic stimulation of these brain areas in a spatially unrelated manner didn’t show any aversion or preference to any scent thereby ruling out possibilities of chance occurrences of the mice behaviour.
This brings us one more step closer to realizing our dreams of instilling false memories in humans. However as wonderful as this technique seems one must not be ignorant of the future ethical ramifications of such a powerful tool.
Memory research has opened up so many exciting new avenues.
There are talks of memories being transferred from one organism to another( So can we transplant Albert Einstein’s or Srinivasa Ramanujan’s genius into an ordinary mortal like me?) as was evidenced by the transfer of memories from a trained to an untrained Aplysia.
There have also been attempts to use Deep Brain Stimulation to create a neural prosthetic for facilitating memory learning and recall in the human hippocampus which was done on epileptic patients who already had electrodes implanted surgically for therapeutic processes. The patients were made to perform neurocognitive tests with electrodes recording activity of CA1 and CA3 regions to determine the circuits involved in recall. Here they used mathematical models of MIMO(multiple input multiple output) from CA3 and CA1 neurons respectively and expressed them in the form of Laguerre-Volterra models that could be solved using Laplace transformation.
Something that looks a bit like this in mathematical language-
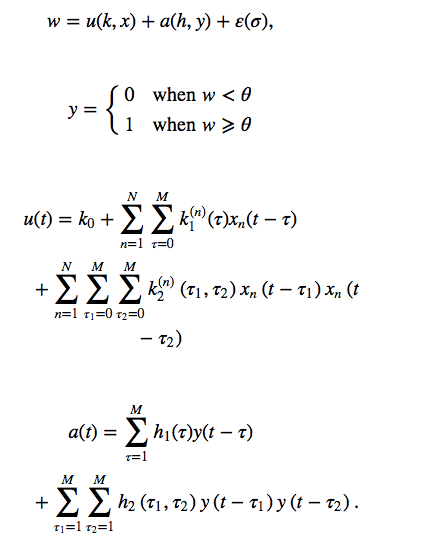
These equations then were decomposed into a number of MISO(Multiple Input Single Output) kernels and then a basic map of the memory circuits involved were developed.
The variables x is the CA3 input and y is the CA1 output while k are the feed forward volterra kernels that describe the relation between the inputs x and the post synaptic potential u. H’s are feed back volterra kernels that describe the relation between preceding y and the after potential a. K0 is the zero order volterra kernels that describes the baseline firing rate, k1 is the first order feed forward volterra kernels that expresses the relation between the nth input Xn to U with respect to the past and present time (t) while the second order k2 expresses the non linear relation between pairs of input spikes and how they along with the preceding order inputs jointly influence U. H1 and H2 are first and second order feed back kernels that describe relation between y taken as a extra input along with respect to time with the after potential a. The prethreshold potential w is expressed as the sum of total post synaptic potential u and after potential a along with a Gaussian noise factor multiplied by the standard deviation sigma. When w crosses the threshold potential theta an action potential is created. However these mathematics though seemingly complicated are actually a very simple representation of our complicated brain architecture.
After mapping the input response neurons in this manner the patients were made to perform neurocognitive tests their performance improved significantly when the output neurons were artificially stimulated by electricity delivered through the electrodes. Tantalizing are the possibilities of such a tool however it’s major disadvantage is it’s invasive nature.
However the advent of optogenetics makes the procedure much less invasive and more accessible with the minute thin optrode being almost similar to a thin catheter being introduced through a minute hole peripherally in the femoral vein into regions as far as the heart hence making these a future possible treatment for neurodegenerative disorders like Alzheimer’s.
Lastly and most shockingly scientists even observed in one study that memories seem to even be transferred to progeny( mice who were trained to be aversive to the cherry blossom acetophenone odour had progeny who also were aversive to the odour) so who even knows what can be the possible future implications where parents may leave their learning and experiences also as a will for their children to inherit which may seem to be far more valuable as compared to material possessions.
Hence I would now like to end this discussion on memory and exciting new techniques to alter it with this quote that sums it all up well- Indeed, memories are not immutable video records of the experienced past that are projected onto a mental theater; they are mnemonic rivers that ebb and flow, and thereby reconstruct the neuronal riverbeds that structurally support various streams of information. With science today having no arbitrary speed limits but being bound only by our own imagination we are now living at an exciting age where we can finally ‘pluck questions from the trees of science fiction and ground them in experimental reality.’
REFERENCES
1)Bliss TV, Lomo T. 1973. Long-lasting potentiation of synaptic
transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J Physiol 232: 331 – 356.
2)Dias, B., Ressler, K. Parental olfactory experience influences behavior and neural structure in subsequent generations. Nat Neurosci 17, 89–96 (2014). https://doi.org/10.1038/nn.3594
3)Guzowski, J. F., McNaughton, B. L., Barnes, C. A., and Worley, P. F. (1999). Environment-specific expression of the immediate-early gene Arc in hip- pocampal neuronal ensembles. Nat. Neurosci. 2, 1120–1124.
4)Guzowski, J. F., Timlin, J. A., Roysam, B., McNaughton, B. L., Worley, P. F.,
and Barnes, C. A. (2005). Mapping behaviorally relevant neural circuits with immediate-early gene expression. Curr. Opin. Neurobiol. 15, 599–606.
5)Hampson RE, Song D, Robinson BS, Fetterhoff D, Dakos AS, Roeder BM, She X, Wicks RT, Witcher MR, Couture DE, Laxton AW, Munger-Clary H, Popli G, Sollman MJ, Whitlow CT, Marmarelis VZ, Berger TW, Deadwyler SA. Developing a hippocampal neural prosthetic to facilitate human memory encoding and recall. J Neural Eng. 2018 Jun;15(3):036014. doi: 10.1088/1741-2552/aaaed7. Epub 2018 Mar 28. PMID: 29589592; PMCID: PMC6576290.
6)Han JH, Kushner SA, Yiu AP, Hsiang HL, Buch T, Waisman A,
Bontempi B, Neve RL, Frankland PW, Josselyn SA. 2009.
Selective erasure of a fear memory. Science 323: 1492 – 1496.
7)Hebb D. 1949. The organization of behavior. Wiley & Sons,
New York.
8)Lammel, S., Lim, B., Ran, C. et al. Input-specific control of reward and aversion in the ventral tegmental area. Nature 491, 212–217 (2012). https://doi.org/10.1038/nature11527
9)Lashley K. 1950. In search of the engram. Symp Soc Exp Biol 4: 454–482.
10)Liu, X., Ramirez, S., Pang, P. et al. Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature 484, 381–385 (2012). https://doi.org/10.1038/nature11028
11)Penfield W, Rasmussen T. 1950. The cerebral cortex of man: A clinical study of localization of function. Macmillan, New York.
12)Ramirez S, Liu X, Lin PA, Suh J, Pignatelli M, Redondo RL, Ryan TJ, Tonegawa S. 2013. Creating a false memory in the hippocampus. Science 341: 387–
13)Ramirez, S., Liu, X., MacDonald, C. et al. Activating positive memory engrams suppresses depression-like behaviour. Nature 522, 335–339 (2015). https://doi.org/10.1038/nature14514
14)Reijmers LG, Perkins BL, Matsuo N, Mayford M. 2007. Localization of a stable neural correlate of associative memory. Science 317: 1230–1233.
15)Sagar, S. M., Sharp, F. R., and Curran, T. (1988). Expression of c-fos protein in brain: metabolic mapping at the cellular level. Science 240, 1328–1331.
16)Sakaguchi M, Hayashi Y. Catching the engram: strategies to examine the memory trace. Mol Brain. 2012 Sep 21;5:32. doi: 10.1186/1756-6606-5-32. PMID: 22999350; PMCID: PMC3462696.
17)Semon R. 1923. Mnemic philosophy. Allen & Unwin, Australia
18)Vetere, G., Tran, L.M., Moberg, S. et al. Memory formation in the absence of experience. Nat Neurosci 22, 933–940 (2019). https://doi.org/10.1038/s41593-019-0389-0