
Circadian rhythms or changes in an organism’s behavior or physiology that follow a cyclical 24 hour pattern are found throughout the tree of life with fungi, plants, insects and mammals all demonstrating them. However when it comes to bacteria, the most ancient of life forms that are ubiquitous dwellers in diverse habitats, the presence of a circadian rhythm was a matter of controversy. For ages bacteria were thought to be clockless owing to the circadian infradian rule assuming that bacteria were unlikely to show 24 hour rhythms as their reproductive cycle spanned less than a day and obviously what use could a clock that ticks longer than your lifespan be?
Research on circadian rhythms in Cyanobacteria-
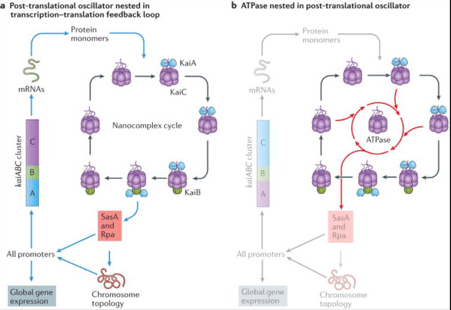
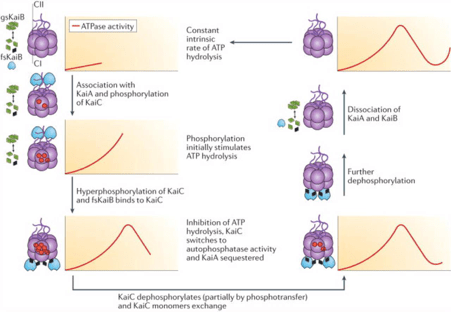
In the 1980’s a series of studies on Cyanobacteria – the oldest photosynthetic organisms present on earth- demonstrated the presence of a circadian clock in them. Many processes in these bacteria are circadian in nature like nitrogen fixation which occurs at midnight 12 hours out of phase from photosynthesis which peaks midday as the enzyme nitrogenase is degraded by even minute quantities of oxygen. (1)Circadian rhythms help anticipate unfavourable environmental conditions in bacteria such as shutting off photosynthetic machinery during the dark hours to avoid unnecessary energy expenditure. The analysis of Cyanobacterial circadian systems was done with an assay know as luciferase reporting assay. The firefly enzyme luciferase that catalysed a reaction in which luciferin is converted oxidatively to oxyluciferin with the emission of light is cloned downstream to the regulatory region of the gene of interest and the resulting vector DNA is introduced into the cells and the cells are grown allowing transcription and translation of the gene products following which they are Lysenko to release the enzyme which when made to react with the substrate luciferin produces a bioluminescence that can be quantified and correlated well with the level of expression of the gene of interest X. (4) Initially the circadian clock was thought to be based on a transcription translation feedback loop(TTFL), however the current understanding of the Cyanobacteria circadian clock is that it revolves around 3 clock proteins known as KaiA, KaiB and KaiC of which Kai C plays a key role. Kai C phosphorylation plays the role of a post translational oscillator which keeps on oscillating between a hypophosphorylated to a hyperphosphorylated state by phosphorylation hand dephosphorylating residues in the C2 domain in the sequence T432 followed by S431 and the phosphotransferase activity functions partially by regenerating ATP from C2 bound ADP and phosphates bound to the residues leading to the clock functioning even at times of low ATP production such as the dark hours of the day. Concomitant with this phosphorylation cycle is the association dissociation of the Kai A KaiB kaiC nanocomplex. Unlike most other phosphorylation reactions this one is exquisitely slow with on,y about 15-16 molecules of ATP being hydrolysed per hour thereby permitting a long 24 hour time constant. (2)Such a biological clock offers immense evolutionary advantage to the bacteria as they outcompete arrhythmic bacteria in a rhythmic environment.(3) Cyanobacteria have hence proven to be the archetypal model for circadian systems in bacteria however what about similar systems in non photosynthetic prokaryotes?
Circadian rhythms in non photosynthetic bacteria-
Before we delve into an account on circadian systems in heterotrophic bacteria it is essential to be introduced to a term call ‘Zeitgeber’. Zeitgeber is a chronobiological term used to refer to any external or environmental cue that serves to entrainment an organism’s biological rhythm to the earth’s 24 hour cycle such as daylight for example.
In an exciting recently published study scientists were successful in identifying circadian clocks in Bacillus subtilis a gram positive non photosynthetic bacillus. The B subtilise genome lacks analogs of Kai proteins that were present in Cyanobacteria but instead have Pern arn’t sim (PAS) domains that are a component of circadian clocks in all eukaryotes and out of 16 such domains ytvA was selected to create a bioluminescent reporter strain. Initially the researchers attempted to check whether light served as a sufficient zeitgeber by applying 12 hour light and 12 hour dark cycles. Pytv::lux expression increased only during the dark phase decreasing in the lit hours. There is an irregular expression of Pytv::lux luciferase with a stable expression occurring only after several days owing to the time needed for the two distinct oscillators the circadian clock and the zeitgeber cycle to form a stable relationship. A free running rhythm occurs which increases in length and eventually gets damped over 5 days when the cultures are subsequently exposed to continuous darkness however when they are grown on glucose depleted media this damping occurs more quickly and abruptly. Findings were validated with yet another PAS gene called KinC that was responsible for sporulation and biofilm formation. Self sustained free running rhythms in the promoter region of ytvA also occurred following entrainment to temperature cycles (12hours at 25.5 degrees Celsius and 12 hours at 28.5 degrees Celsius) in contrast to the light entrainment here surprisingly there was a higher level of expression during the warm phase as compared to the cold phase and the free running period was greatly suppressed by the presence of glucose and glycerol in the culture media in stark contrast to what was observed in the light entrained cultures.
This shows that the clock is sensitive to environmental changes like the carbon sources. Also all cultures showed biofilm formation demonstrated by a visible pellicle.
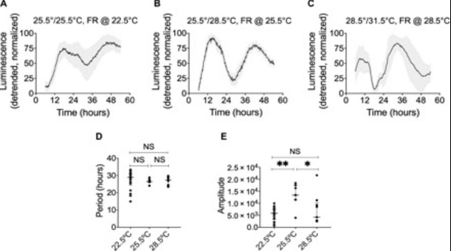
Another hallmark of these clocks is that they have a Q10 value close to 1 and show temperature compensation which means that the free running periods across a 10 degree temperature range(6 degrees in actual practice) was not very different. The scientists also determined the effect of varying the entrainment magnitude on the phase of entrainment using temperature as the zeitgeber and found that the phase of the first cycle of oscillation was later at lower temperatures and earlier at higher temperatures. However one doubt that may occur to you is how can we be so certain that these cycles are truly reflective of a circadian rhythm and are not a mere consequence of environmental changes(masking)? To differentiate between these different T cycles were used.
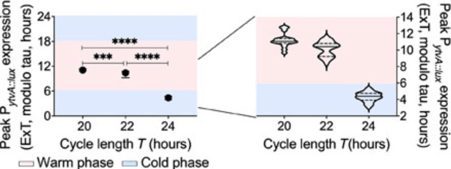
T is the duration of the entire entertaining cycles and for earth T is 24 hours. For a circadian clock to be true it needs to show synchrony between the zeitgeber cycle and the rhythm. A longer running free period will entrain to a later phase in a 24h cycle than one with a shorter period. It can hence be deduced that a given individual with a set circadian rhythm will entrain earlier in a shorter cycle and later in a longer cycle while a masking signal simply synchronises to the same phase irrespective of the length of the zeitgeber cycle. B subtilis was found to entrain later as T cycle became shorter providing evidence of a robust circadian system that interprets the zeitgeber cycle as an oscillator rather than simply responding to environmental changes as a switch does.
The calculation of the free running period was done using a non linear model as described below and was analysed with R programming language .(5)
𝑓(𝑡)=𝑎0·𝑒−𝑘0·𝑡+𝑎1·𝑒−𝑘1·𝑡·cos(2π·(𝑡−θ)𝑇)
t= time in hours
a0 = max amplitude of baseline signal
K0= rate of decay of baseline signal
a1=amplitude of oscillation
K1= rate of decay of oscillation
T= period of oscillation
Theta= phase of signal at start of experiment
Clinical Implications of this discovery
So what if bacteria possess circadian rhythms? How does it affect us in any way? Interestingly the account describes above regarding clocks in B subtilis was not the first to be reported a,one non photosynthetic bacteria. Few years back scientists discovered such a clock in Enterobacter aerogenes ( now known as Klebsiella aerogenes ) that exists as a commensal in the human gut. Based on an observation that K aerogenes showed rapid proliferation in the presence of physiological levels of human melatonin that are usually seen in the gut( gut enterochromaffin cells, cholangiocytes etc do contain melatonin synthesis enzymes apart from pineal gland) scientists performed luciferase assay based investigations on this bacterium and showed that these organisms showed robust circadian rhythms (in motA motility protein activity) in 31-44% of the cultures.
While melatonin didn’t affect the circadian rhythm per se it did affect the phase of peak bioluminescence which showed high variability among different replicates in the absence of melatonin that became highly synchronised on melatonin application.
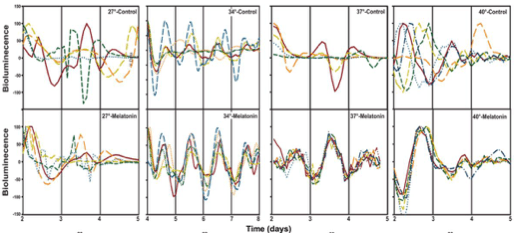
So the bacteria is in essence a meta organism with its own endogenous clock responding to the host’s clock driven rhythm(6). Studies in mice have shown that loss of the host’s clock genes(7), a high fat diet(8), changes in feeding time and jet lag(9) all have an impact on the rhythmicity of gut microbiota and alter the composition to favour development of obesity and insulin resistance. In fact another study found that disruption of the rhythmicity of 13 microbial taxa were seen in patients with type 2 diabetes mellitus.(10) This could be due to alteration in metabolites produced by the bacteria. Mice with regular diets exhibited daily oscillation in butyrate levels- a compound that plays a key role in intestinal homeostasis and produced by gut micro biome while those on a high fat diet lost this oscillation and were prone to develop obesity.(11)

In fact mice on HFD could be protected from obesity by Time Restricted Feeding even without reducing calorie intake(12) Doesn’t this sound familiar? Living as we are in the era of diets like intermittent fasting and Time Restricted Feeding it could indeed be possible that the endogenous rhythms of gut microbes have a role to play in the beneficial effect of such diets. The role of microbes in IF has been demonstrated by several studies including one which showed increased mitochondrial synthesis in hippocampus and improved synapse ultra structure leading to reversal of cognitive decline due to DM.(13) In fact the gut micro biome is also shown to cause proper alignment of circadian rhythms in hosts on IF thereby holding promise for treating disorders of circadian rhythm like sleep disorders. (14)Also the very fact that microbes have an endogenous clock could also potentially be used to time the administration of antibiotics to the metabolically active phase of the bacterial clock so as to maximise their efficacy. The discovery of bacterial clocks opens up new exciting avenues of research, to determine how best we can synchronise the ticking of their clocks to our own, in order to maximise our health and well being.
REFERENCES
(1) Mitsui A, et al. Strategy by which nitrogen-fixing unicellular cyanobacteria grow photoautotrophically. Nature. 1986; 323:720–722.
(2) Johnson CH, Zhao C, Xu Y, Mori T. Timing the day: what makes bacterial clocks tick? Nat Rev Microbiol. 2017 Apr;15(4):232-242. doi: 10.1038/nrmicro.2016.196. Epub 2017 Feb 20. PMID: 28216658; PMCID: PMC5696799.
(3) Woelfle MA, Ouyang Y, Phanvijhitsiri K, Johnson CH. The adaptive value of circadian clocks: an experimental assessment in cyanobacteria. Curr Biol. 2004; 14:1481–1486. [PubMed: 15324665]
(4) Kondo T, Strayer CA, Kulkarni RD, et al. Circadian rhythms in prokaryotes: luciferase as a reporter of circadian gene expression in cyanobacteria. Proc Natl Acad Sci U S A. 1993;90(12):5672-5676. doi:10.1073/pnas.90.12.5672
(5) Eelderink-Chen Z, Bosman J, Sartor F, Dodd AN, Kovács ÁT, Merrow M. A circadian clock in a nonphotosynthetic prokaryote. Sci Adv. 2021;7(2):eabe2086. Published 2021 Jan 8. doi:10.1126/sciadv.abe2086
(6) Paulose JK, Wright JM, Patel AG, Cassone VM. Human Gut Bacteria Are Sensitive to Melatonin and Express Endogenous Circadian Rhythmicity. PLoS One. 2016;11(1):e0146643. Published 2016 Jan 11. doi:10.1371/journal.pone.0146643
(7) Liang X, Bushman FD, FitzGerald GA. Rhythmicity of the intestinal microbiota is regulated by gender and the host circadian clock. Proc Natl Acad Sci U S A. 2015;112(33):10479-10484. doi:10.1073/pnas.1501305112
(8)Zarrinpar A, Chaix A, Yooseph S, Panda S. Diet and feeding pattern affect the diurnal dynamics of the gut microbiome. Cell Metab. 2014;20(6):1006-1017. doi:10.1016/j.cmet.2014.11.00
(9) Thaiss CA, Zeevi D, Levy M, et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell. 2014;159(3):514-529. doi:10.1016/j.cell.2014.09.048
(10) Reitmeier S, Kiessling S, Clavel T, et al. Arrhythmic Gut Microbiome Signatures Predict Risk of Type 2 Diabetes. Cell Host Microbe. 2020;28(2):258-272.e6. doi:10.1016/j.chom.2020.06.004
(11) Leone V, Gibbons SM, Martinez K, et al. Effects of diurnal variation of gut microbes and high-fat feeding on host circadian clock function and metabolism. Cell Host Microbe. 2015;17(5):681-689. doi:10.1016/j.chom.2015.03.006
(12) Hatori M, Vollmers C, Zarrinpar A, et al. Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice fed a high-fat diet. Cell Metab. 2012;15(6):848-860. doi:10.1016/j.cmet.2012.04.019
(13) Liu Z, Dai X, Zhang H, et al. Gut microbiota mediates intermittent-fasting alleviation of diabetes-induced cognitive impairment. Nat Commun. 2020;11(1):855. Published 2020 Feb 18. doi:10.1038/s41467-020-14676-4
(14) Daas MC, de Roos NM. Intermittent fasting contributes to aligned circadian rhythms through interactions with the gut microbiome. Benef Microbes. 2021;12(2):147-161. doi:10.3920/BM2020.0149
